
Design, synthesis and anti-breast cancer of sonosensitizers for synergistic sonodynamic therapy and NF-κB transcription inhibition
As a complement to chemotherapy, sonodynamic therapy (SDT) has been recognized as a spatiotemporal and non-invasive cancer treatment modality. During SDT treatment, a sonosensitizer is used to absorb low-intensity ultrasound, converting it into reactive oxygen species (ROS), resulting in damage to tumor cells and tissues. In addition to specific spatiotemporal selectivity, SDT can also penetrate deep tissues with the help of ultrasound to radically treat deep tumor tissues, which has great clinical transduction potential.
Recently, Professor Li Yuanyuan of Chen Xin's research group of Wuhan University of Light Industry and Professor Zhang Silong and Professor He Huan of Wuhan University of Science and Technology collaborated in the Journal of Medicinal Chemistry, a top journal in medicinal chemistry In this article, it was reported that the resveratrol backbone was used as the target part of NF-κB, and the electron donor (triphenylamine) and electron acceptor (benzothiazole) conjugated backbone were linked, and a new sonosensitizer was constructed using the design concept of bifunctional molecules, and the effects of synergistic sonodynamic therapy and NF-κB transcriptional inhibition in anti-breast cancer were studied.
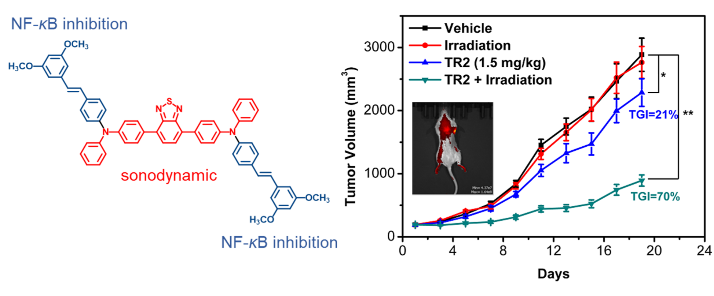
Figure 1. Structure and anti-breast cancer effect of the new sonosensitizer
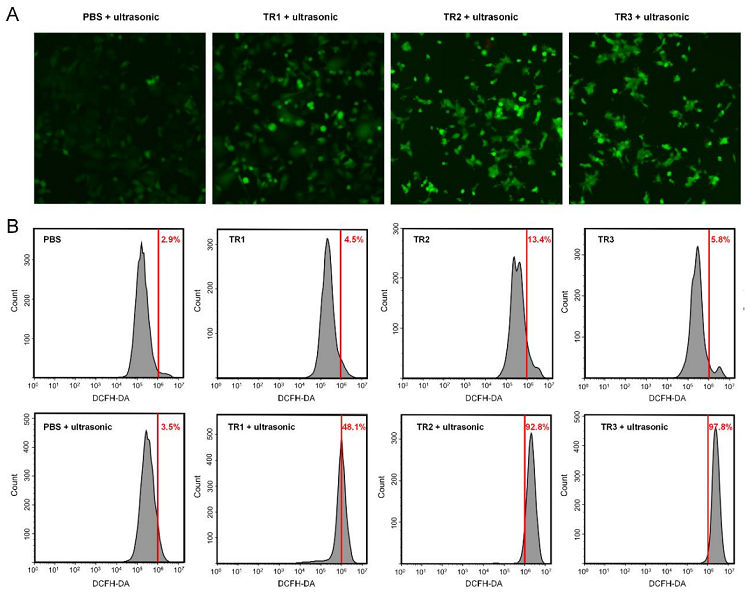
Studies have shown that the sonosensitizer can target NF-κ B, inhibit the activity of NF-κB by absorbing low-intensity ultrasound into reactive oxygen species, and achieve anti-breast cancer effect. The authors validated the acoustic dynamic efficacy of the novel sonosensitizer in MCF-7 human breast cancer cells. Cells are incubated in 10.0 μM sonosensitizer for 4 h followed by 0.8 W/cm2Ultrasonic treatment for 30 sec, followed by treatment with fluorescent indicator DCFH-DA. After the addition of the sonosensitizer, DCFH-DA fluorescence is clear in cells, staining cells up to 978%, and cell ROS volume increases. At the same time, the novel sonosensitizer showed good NO inhibitory effect on RAW 264.7 macrophages, and the effective concentration of half reached 68.9 nM. In addition, the results of their in vitro anticancer activity test showed that these sonosensitizers showed specific toxicity to MCF-1 cells at doses greater than 0.7 μM, which could cause inhibition of the NF-κBpathway, and TR2 and TR3 were superior to TR7 in inhibiting MCF-1 cell proliferation.
Figure 2. Effect of novel sonosensitizers on ROS generation in vitro
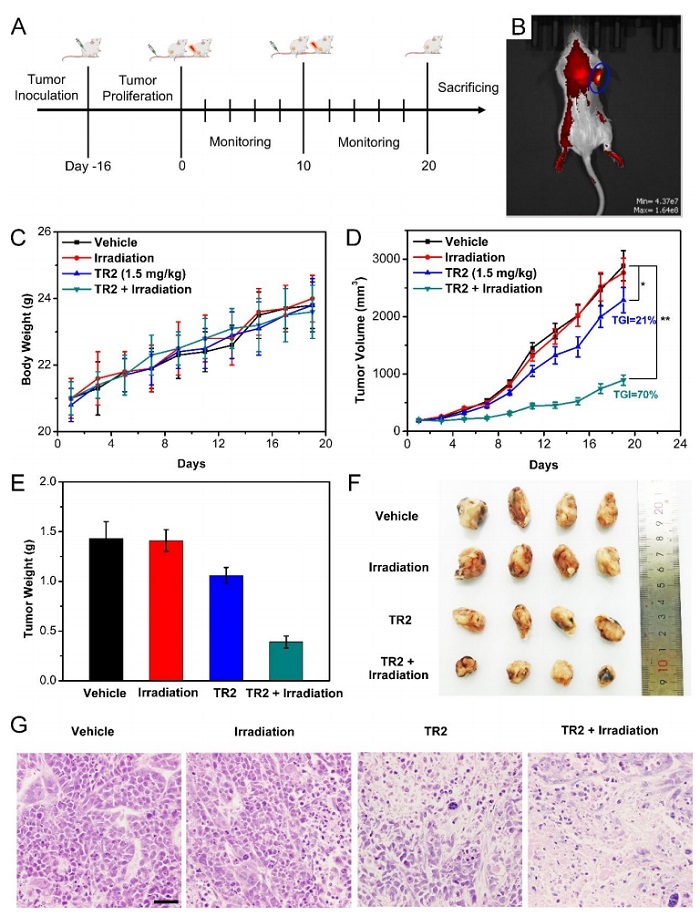
Finally, the authors further evaluate the antitumor efficacy of TR2 in a xenograft mouse model. During the entire study, the mice tolerated TR2 well and did not see significant weight loss, proving the safety of ultrasound. After intravenous administration of TR2, tumor growth in mice was significantly lower than in the control group (TGI = 21%), which may be associated with inhibition of cancer cell NF-κ B related. In addition, cytokine levels in the mice serum decreased by 42% compared to the control group, indicating that TR2 induced an anti-inflammatory response. Notably, the combination of TR2 and ultrasound increased tumor growth inhibition to 70%. Hematoxylin-eosin staining of tumor sections further confirmed that TR2 combined with ultrasound group could significantly inhibit the growth of MCF-7 cells and had good anti-cancer effects. At the same time, no obvious changes in cell structure and organ damage were seen in all major organs (heart, liver, spleen, lung, kidney), indicating that the measured dose of acoustic sensitizer had low toxicity.
Figure 3. Study of novel sonosensitizers against breast cancer in mice
In summary, using the resveratrol backbone as a targeted portion of NF-κB, the authors successfully designed a safe anti-breast cancer treatment strategy for synergistic sonodynamic therapy and NF-κBinhibition. The authors' work provides valuable therapeutic advantages for the further development of SDT sonosensitizers.
Name: Eric Yuan
Mobile:86-18427358861
Tel:86-18427358861
Whatsapp:+86 18427358861
Email:yihekeji@chembuying.com
Add: